... an economical use of fertilizers requires that they merely supplement the natural supply in the soil, and that the latter should furnish the larger part of the soil material used by the crop.
—T.L. Lyon and E.O. Fippin, 1909
Both nitrogen and phosphorus are needed by plants in large amounts, and both can cause environmental harm when present in excess. They are discussed together in this chapter because we don’t want to prioritize the management of one nutrient and neglect the other; it’s important to consider balanced nutrition. And when applying manures and composts, which contain N and P (as well as other nutrients, of course), there is no alternative to taking both into consideration. Nitrogen losses are an economic concern for farmers: If not managed properly, a large fraction (as much as half in some cases) of applied N fertilizer can be lost instead of used by crops. Environmental concerns with N include the leaching of soil nitrate to groundwater, excess N in runoff, and losses of nitrous oxide (a potent greenhouse gas). For P, the main concerns are losses to freshwater bodies through runoff and leaching into tile drains.
High-nitrate groundwater is a health hazard to infants and young animals because it decreases the blood’s ability to transport oxygen. There is accumulating evidence that high-nitrate drinking water might have adverse health effects on adults as well. In addition, as surface waters become enriched with nutrients (the process is called eutrophication) there is an increase in aquatic plant growth. Nitrate stimulates the growth of algae and aquatic plants, just as it stimulates the growth of agricultural plants. The growth of plants in many brackish estuaries and saltwater environments is believed to be limited by a lack of N. So, undesirable microorganisms flourish when nitrate leaches through soil or runs off the surface and is discharged into streams, eventually reaching water bodies like the Gulf of Mexico, the Chesapeake Bay, Puget Sound or the Great Lakes, and increasingly many others around the world. In addition, the algal blooms that result from excess N and P cloud water, blocking sunlight to important underwater grasses that are home to numerous species of young fish, crabs and other bottom dwellers. The greatest concern, however, is the dieback of the algae and other aquatic plants. These plants settle on the bottom of the affected estuaries, and their decomposition consumes dissolved oxygen in the water. The result is an extended area of very low oxygen concentrations in which fish and other aquatic animals cannot live. This is a serious concern in many estuaries around the world, and despite government efforts to curtail the flow of nutrients, most of these dead zones appear to be growing rather than shrinking (the Gulf of Mexico’s dead zone still averages three times larger than the goal set by the U.S. Environmental Protection Agency).
Nitrogen can also be lost from soil by denitrification, a microbial process that occurs primarily when soils are saturated with water. It is especially problematic in soils with poor structure due to compaction or other causes, frequently a result of excessive tillage. Soil bacteria convert nitrate to both nitrous oxide (N2O) and N2. While N2 (two atoms of nitrogen bonded together) is the most abundant gas in the atmosphere and not of environmental concern, each molecule of N2O gas—largely generated by denitrification, with some contribution from nitrification—has approximately 300 times more climate change impact than a molecule of carbon dioxide. According to the U.S. Environmental Protection Agency, N2O accounts for 55% of the agricultural greenhouse gas emissions and 5% of the total emissions of all economic sectors combined, which is equivalent to twice the impact from the aviation industry.
Phosphorus losses from farms are generally small in relation to the amounts present in soils. However, small quantities of P loss have great effects on water quality because P is the nutrient that frequently limits the growth of freshwater aquatic weeds, algae and cyanobacteria (also called “blue green algae”). Phosphorus damages the environment when excess amounts are added to a lake from human activities (agriculture, rural home septic tanks, or urban sewage and street runoff). This eutrophication increases algae growth, which makes fishing, swimming and boating unpleasant or difficult. When excess aquatic organisms die, decomposition removes oxygen from water and leads to fish kills. This is a large concern in the freshwater lakes near the authors’ homes in Vermont and New York where dairy farming is prevalent, and in recent years it has created a very extensive low oxygen (hypoxia) zone in the western part of Lake Erie.
All farms should work to have the best N and P management possible for economic as well as environmental reasons. This is especially important near bodies of water that are susceptible to accelerated weed or algae growth. However, don’t forget that nutrients from farms in the Upper Midwest are contributing to problems in the Gulf of Mexico over 1,000 miles away.
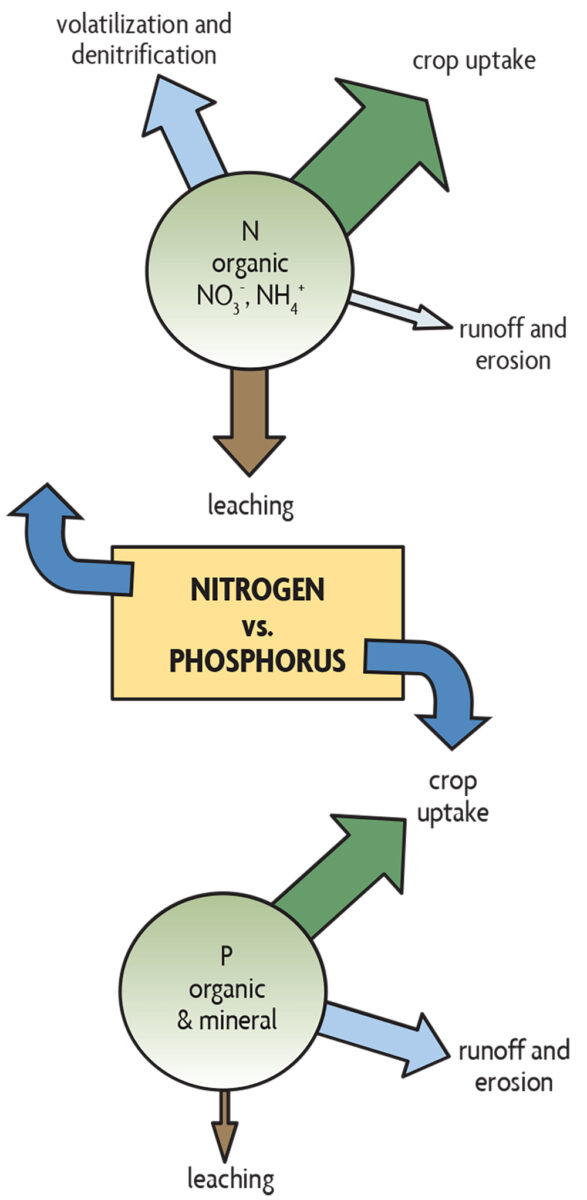
There are major differences between the way N and P behave in soils (Figure 19.1, Table 19.1). Both N and P can, of course, be supplied in applied fertilizers. But aside from legumes that can produce their own N because of the bacteria living in root nodules, crop plants get their N from decomposing organic matter. On the other hand, there is no biological process that can add P to soils, and plants get their P from soil minerals as well as from decomposing organic matter. Nitrate, the primary form in which plants absorb nitrogen from the soil, is very mobile in soils, while P movement in soils is very limited.
Most N loss from soils occurs when nitrate leaches, is converted into gases by the process of denitrification, or is volatilized from surface ammonium. When water exceeds plant needs, large amounts of nitrate may leach from sandy soils, while denitrification is generally more significant in heavy loams and clays. On the other hand, P is lost from soils in lesser quantities when it is carried away in runoff or in sediments eroded from fields, construction sites and other exposed soil (see Figure 19.1 for a comparison between relative pathways for N and P losses). But generally lower P losses are associated with higher impact per unit of nutrient on water quality, so the overall environmental concerns with both N and P are therefore significant. Except for highly manured fields, P losses in runoff and erosion from healthy grasslands is usually quite low because both runoff water and sediment loss are very low. Phosphorus leaching is a concern in fields that are artificially drained. With many years of excessive manure or compost application, soils saturated with P (often sands with low P sorption capacity) can start leaking P with the percolating water and can discharge it through drain lines or ditches. Also, liquid manure can move through preferential flow paths (wormholes, root holes, cracks, etc., especially in clay soils) directly to subsurface drain lines and contaminate water in ditches, which is then discharged into streams and lakes (see also Chapter 17). Cover crops help lessen nutrient loss by preferentially filling many of the large continuous pores with roots, causing more water to flow through the main matrix of the soil and allowing for better nutrient retention.
PROBLEMS USING EXCESS N FERTILIZER
There are quite a few reasons you should not apply more N than is needed by crops. N fertilizers are costly, and many farmers are judicious with application rates. However, there are other problems associated with using more N than needed: 1) groundwater and surface water become polluted with nitrates; 2) more N2O (a potent greenhouse gas and source of ozone depletion) is produced during denitrification in soil; 3) a lot of energy is consumed in producing N, so wasting N is the same as wasting energy; 4) using higher N than needed is associated with accelerated decomposition and loss of soil organic matter; and 5) very high rates of N are frequently associated with high levels of insect damage. For many farmers, the challenge is knowing the correct N fertilizer rate for their crop in the particular growing season. With this uncertainty and with the risk of yield losses from insufficient fertilizer applications, they tend to apply more than needed in many years. Good N management tools can help address this concern.
Improving N and P management can help reduce reliance on commercial fertilizers. A more ecologically based system, with good rotations, reduced tillage and more active organic matter, should provide a large proportion of crop N and P needs. Better soil structure and attention to the use of appropriate cover crops can lessen N and P loss by reducing leaching, denitrification and/or runoff. Reducing the loss of these nutrients is an economic benefit to the farm and, at the same time, an environmental benefit to society. The greater N and P availability may be thought of as a fringe benefit of a farm with an ecologically based cropping system.
In addition, the manufacture, transportation and application of N fertilizers are very energy intensive. Of all the energy used to produce corn (including the manufacture and operation of field equipment), the manufacture and application of N fertilizer represents close to 30%. In the late 2010s energy (and N fertilizer) costs decreased from their record high levels, but it still makes sense for both environmental and economic reasons to use N fertilizers wisely. Relying more on biological fixation of N and efficient cycling in soils reduces depletion of a nonrenewable resource and may save you money as well. Although P fertilizers are less energy consuming to produce, a reduction in their use helps preserve this nonrenewable resource—the world’s P mines will run out at some time in the future.
| Table 19.1 Comparing Soil N and P | |
|---|---|
| Nitrogen | Phosphorus |
| Nitrogen becomes available from decomposing soil organic matter, commonly supplying about one third or more of crop uptake. N is mostly available to plants as nitrate (NO3–), a form that is very mobile in soils. Some ammonium (NH4+) and small nitrogen-containing organic molecules such as amino acids are also taken up by plants. Nitrate can be easily lost in large quantities by leaching to groundwater or by conversion to gases (N2, N2O). Nitrogen can be added to soils by biological N fixation (legumes). Cover crops can store nitrogen that would otherwise be lost by leaching and denitrification, providing the N to the following crop. | Phosphorus becomes available from decomposing soil organic matter and minerals. P is relatively immobile and is only available to plants in small concentrations as dissolved phosphorus in the soil solution, mainly as H2PO4– and HPO4–2. P is mainly lost from soils through runoff and erosion. However, excessive fertilizer P or manure application on well-structured soils and on those with tile drainage has resulted in P loss to drainage water. No equivalent reaction can add new P to soil, although many bacteria and some fungi (especially mycorrhizae) help plants take up more P. Cover crops can mobilize P from soil and store it in their tissue, providing extra P to the following crop. |
Management of N and P
Nitrogen and phosphorus behave very differently in soils, but many of the management strategies are actually the same or very similar. They include the following:
- Take all nutrient sources into account.
- Estimate nutrient availability from all sources.
- Use soil tests to assess available nutrients. (Nitrogen soil tests are not available for all states. Some make N fertilizer recommendations based on fertilizer trials and estimates of cover crop contributions. Other methods for making N recommendations are discussed later in this chapter.)
- Use manure and compost tests to determine nutrient contributions.
- Consider nutrients in decomposing crop residues (for N only).
- Reduce losses and enhance uptake (use 4R-Plus principles, fertilizer application using the right rate, at the right time, in the right place, in the right amount, plus conservation practices; see Chapter 18).
- Use nutrient sources more efficiently.
- Use localized placement of fertilizers below the soil surface whenever possible.
- Split fertilizer application if leaching or denitrification losses are a potential problem (almost always for N only).
- Apply nutrients when leaching or runoff threats are minimal.
- Reduce tillage.
- Use cover crops.
- Include perennial forage crops in rotation.
- Balance farm imports and exports once crop needs are being met.
Cover crops combined with minimal or no tillage is a set of practices that work well together. They improve soil structure; reduce the loss of nutrients through leaching, runoff and erosion; reduce denitrification loss of nitrates; and tie up N and P that otherwise might be lost between cash crops by storing these nutrients in organic forms.
Estimating Nutrient Availability
Good N and P management practices take into account the large amount of plant-available nutrients that come from the soil, especially soil organic matter and any additional organic sources like manure, compost, or a rotation crop or cover crop. Fertilizer should be used only to supplement the soil’s supply in order to provide full plant nutrition (Figure 19.2). Organic farmers try to meet all demands through these soil sources because additional organic fertilizers are generally very expensive. This is typically done by incorporating a legume as a crop or cover crop into the rotation or by adding high-N organic nutrient sources. When using organic fertilizers, the higher the percent N in the compost or in the other material, the more N will become available to plants. Little to no N will be available to plants if the amendment is around 2% N or less (corresponding to a high C:N ratio). But if it’s around 5% N, about 40% of the N in the amendment will be available. And if it’s 10% or 15% N (corresponding to a very low C:N ratio), 70 percent or more of the N in the amendment will be available to crops. On integrated crop-livestock farms soil organic N and P sources are typically sufficient to meet the crop’s demand, but not always.
Since most plant-available P in soils is relatively strongly adsorbed by organic matter and clay minerals, estimating P availability is routinely done through soil tests. The amount of P extracted by chemical soil solutions can be compared with results from crop response experiments and can provide good estimates of the likelihood of a response to P fertilizer additions, which we discuss in Chapter 21.
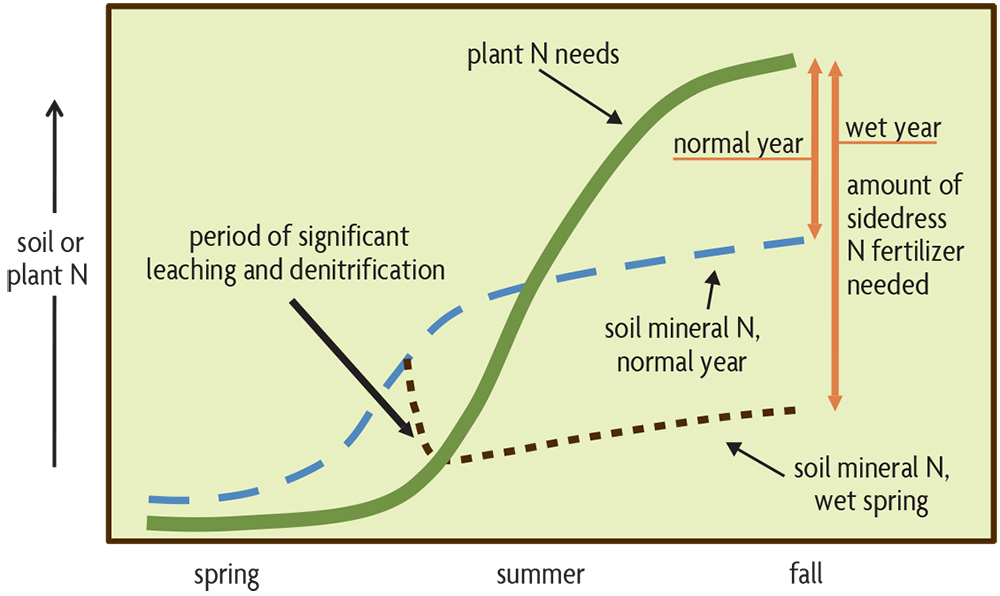
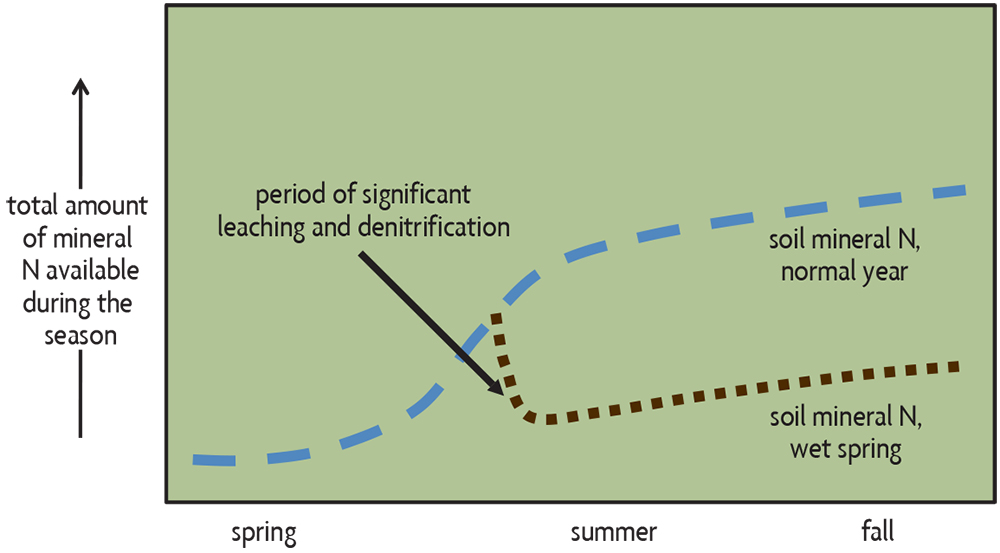
Estimating N fertilizer needs is more complex, and soil tests generally cannot provide all the answers. The primary reason is that the amounts of plant-available N, mostly nitrate, can fluctuate rapidly as organic matter is mineralized and as N is lost through leaching or denitrification. These processes are greatly dependent on soil organic matter contents, additional N contributions from organic amendments, and weather-related factors like soil temperature (higher temperatures increase N mineralization) and soil wetness (saturated soils cause large leaching and denitrification losses, especially when soils are warm). Mineral forms of N begin to accumulate in soil in the spring but may be lost by leaching and denitrification during a very wet period (Figure 19.2). When plants germinate in the spring, it takes a while until they begin to grow rapidly and take up a lot of N (Figure 19.3). Weather affects the required amount of supplemental N in two primary ways. In years with unusually wet weather in the spring, an extra amount of sidedress (or topdress) N may be needed to compensate for relatively high mineral N loss from soil (Figure 19.3). The increasing rainfall intensity in some regions makes the use of sidedress N even more important. Research on corn in Minnesota from 2015 to 2019—where 75 percent of the sites evaluated had one month during the growing season with 150 percent of normal rainfall—indicated using sidedress N with some N applied before planting didn't decrease yields and actually increased yields by an average of 11 bushels an acre in a quarter of situations.
On the other hand, in dry years, especially drought spells during the critical pollination period, yields will be reduced, and the N uptake and needed N fertilizer are therefore lower (not shown in Figure 19.3). However, you really don’t know at normal sidedress time whether there will be a drought during pollination, so there is no way to adjust for that. For a field with a given soil type and set of management practices, the actual amount of required N also depends on the complex and dynamic interplay of crop growth patterns with weather events, which are difficult to predict. In fact, optimum N fertilizer rates for corn without organic amendments in the U.S. corn belt have been found to vary from as little as 0 pounds per acre to as much as 250 pounds per acre. Those are the extremes, but, nevertheless, it is a great challenge to determine the optimum economic N rate. There may be different issues arising in other regions. In the Northwest’s maritime region, large amounts of winter rainfall normally result in very low levels of available N in spring. Without much year-to-year carryover of mineral N and with low organic matter decomposition during the cool season, it is especially important to be sure that some readily available N is near the developing seedling of spring planted crops.
Fixed and Adaptive Methods for Estimating Crop N Needs
Several approaches are used to estimate crop N needs, and they can be grouped into fixed and adaptive approaches. Fixed (static) approaches assume that the N fertilizer needs do not vary from one season to another based on weather conditions, which may work well in drier climates but are very imprecise in a humid climate. Adaptive methods recognize that precise N fertilization requires additional data from field samples, sensors or computer models to modify the N rate for a particular production environment.
The mass-balance approach, a fixed approach, is the most commonly used method for estimating N fertilizer recommendations. It is generally based on a yield goal and associated N uptake, minus credits given for non-fertilizer N sources such as mineralized N from soil organic matter, preceding crops and organic amendments. However, studies have shown that the relationship between yield and optimum N rate is very weak for humid regions. While higher yields do require more N, the weather pattern that produces higher yields also implies 1) that larger and healthier root systems can take up more soil N, and 2) that frequently the weather pattern stimulates the presence of higher levels of nitrate in the soil. Conversely, very wet conditions cause reduced yields due to insufficient soil aeration and low soil N availability.
Several leading U.S. corn-producing states have adopted the maximum return to N (MRTN) approach, another fixed method that largely abandons the mass-balance approach. It provides generalized recommendations based on extensive field trials, model-fitting and economic analyses. It is only available for corn at this time. The rate with the largest average net return to the farmer over multiple years is the MRTN, and the recommendations vary with grain and fertilizer prices. Adjustments based on realistic yield expectation are sometimes encouraged. The MRTN recommendations are based on comprehensive field information, but owing to generalizing over large areas and over many seasons, it does not account for the soil and weather factors that affect N availability and is therefore inherently imprecise for an individual field.
The adaptive approaches, described in the following paragraphs, attempt to take into account seasonal weather, soil type and management effects, and require some type of measurement or model estimate during the growing season.
The pre-sidedress nitrate test (PSNT) measures soil nitrate content in the surface layer of 0–12 inches and allows for adaptive sidedress or topdress N applications. It implicitly incorporates information on early season weather conditions (Figure 19.2) and is especially successful in identifying N-sufficient sites: those that do not need additional N fertilizer. It requires a special sampling effort during a short time window in late spring, and it is sensitive to timing and mineralization rates during the early spring. The PSNT is usually called the late spring nitrate test (LSNT) in the midwestern United States.
Pre-plant nitrate and labile N tests measure soil nitrate, soil nitrate plus ammonium, or readily available organic nitrogen in the soil early in the season to guide N fertilizer applications at planting. These approaches are more effective in drier climates, like in the U.S. Great Plains where seasonal gains of inorganic forms of N are more predictable and losses from leaching or denitrification are generally minimal. Fall soil sampling can provide valuable information for N management for winter wheat while early spring season sampling is preferable for evaluating N needs for corn. These approaches cannot incorporate the seasonal weather effects, as the samples are analyzed prior to the growing season, which inherently limits its precision compared to the PSNT. Recent advances in crop sensing and modeling allow adaptive approaches based on seasonal weather and local soil variation. Leaf chlorophyll meters that measure light transmission in leaves and satellite, aerial, drone or tractor-mounted sensors that determine light reflection from leaves are used for assessing leaf or canopy N status and biomass, which can then guide sidedress N applications. Environmental information systems and dynamic simulation models are now also being employed for N management, with successful applications for wheat and corn. This approach takes advantage of increasingly sophisticated environmental databases, such as radar-based, high-resolution precipitation estimates and detailed soil databases, and can be used to provide input information for computer models. We discuss these further in Chapter 21.
Evaluation at the End of the Season
To evaluate the success of a fertility recommendation, farmers sometimes plant field strips with different N rates and compare yields at the end of the season. This can be done for vegetable crops as well as for crops like grain corn. Another option is to sample for soil nitrate after harvest, sometimes called a “report card” assessment, to evaluate residual levels of available N. The lower stalk nitrate test is also sometimes used to assess, after the growing season, whether corn N rates were approximately right or too low or too high. These methods are neither fixed nor adaptive approaches for the current year, since evaluation is made at the end of the season, but they may help farmers make changes to their fertilizer application rates in following years. Adaptive management may therefore also include farmer-based experimentation and adjustment to local conditions.
Planning for N and P Management
Although N and P behave very differently in soils, the general approaches to their management are similar (Table 19.2). The following considerations are important for planning management strategies for N and P.
| Table 19.2 Comparison of N and P Management Practices | |
|---|---|
| Nitrogen | Phosphorus |
| Use fixed-rate approaches for planning purposes and adaptive approaches to achieve precision. | Test soil regularly (and follow recommendations). |
| Test manures and credit their N contribution. | Test manures and credit their P contribution. |
| Use legume forage crops in rotation and/or legume cover crops to fix N for following crops, and properly credit legume N contribution to following crops. | No equivalent practice is available (although cover crop and cash crop mycorrhizae help mobilize soil P already there, making it more available to plants). |
| Time N applications as close to crop uptake as possible, and place to reduce runoff or gaseous losses. | Time and place P application to reduce runoff potential. |
| Reduce tillage in order to leave residues on the surface and to decrease runoff and erosion. | Reduce tillage in order to leave residues on the surface, to decrease runoff and erosion, and to keep mycorrhizal network intact. |
| Use sod-type forage crops in rotation to reduce nitrate leaching and runoff, making N more available to following crops. | Use sod-type forage crops in rotation to reduce the amount of runoff and erosion losses of P, making P more available to the following crop. |
| Use grass cover crops, such as cereal rye, to capture soil nitrates leftover following the economic crop. | Use grass cover crops, such as cereal rye, to protect soil against erosion. |
| Make sure that excessive N is not coming onto the farm (biological N fixation plus fertilizers plus feeds). | After soil tests are in the optimal range, balance the farm’s P flow (don’t import much more onto the farm than is being exported). |

Credit nutrients in manures, rotation crops, decomposing sods, cover crops and other organic residues. Before applying commercial fertilizers or other off-farm nutrient sources, you should properly credit the various on-farm sources of nutrients. In some cases, there is more than enough fertility in the on-farm sources to satisfy crop needs. If manure is applied before sampling soil, the contribution of much of the manure’s P and all its potassium will be reflected in the soil test. The pre-sidedress nitrate test can estimate the N contribution of the manure (see Chapter 21 for a description of N soil tests). The only way to really know the nutrient value of a particular manure is to have it tested for its fertilizer value before applying it to the soil; many soil test labs also analyze manures. (Although a manure analysis test is recommended and will provide the most accurate result, estimates can be made based on average manure values, such as those given in Table 12.1.)
Because significant ammonia N losses can occur in as little as one or two days after application, the way to derive the full N benefit from surface-applied manure (or urea for that matter) is to incorporate it as soon as possible. Much of the manure N made available to the crop is in the ammonium form, and losses occur as some is volatilized as ammonia gas when manures dry on the soil surface. A significant amount of the manure’s N may also be lost when application happens a long time before crop uptake occurs. Even if incorporated, about half of the N value of a fall manure application may be lost by the time it is needed by the crop in the following year.
MANURE APPLICATION, TILLAGE AND N LOSS
When using some tillage, it makes sense to incorporate manure as soon after application as weather and competing work priorities allow. With no-till there are low-disturbance manure injectors that place liquid manure in the soil with minimal N loss.
Legumes, either as part of rotations or as cover crops, and well-managed grass sod crops can add N to the soil for use by the next crop (Table 19.3). Nitrogen fertilizer decisions should take into account the amount of N contributed by manures, decomposing sods and cover crops. If you correctly fill out the form that accompanies your soil sample, the recommendation you receive may take these sources into account. However, not all soil testing labs do that; most do not even ask whether you’ve used a cover crop. If you can’t find help deciding how to credit nutrients in organic sources, take a look at chapters 10 (cover crops), 11 (rotations) and 12 (animal manures, discussed as part of integrated livestock-cropping systems). Also, some of the adaptive simulation models described above can incorporate such credits into recommendations, while also accounting for variable weather conditions. For an example of crediting the nutrient value of manure and cover crops, see the section “Making Adjustments to Fertilizer Application Rates” in Chapter 21.
| Table 19.3 Examples of Nitrogen Credits for Previous Crops | |
|---|---|
| Previous crop | N credits (pounds per acre) |
| Corn and most other crops | 0 |
| Soybeans2 | 0–40 |
| Grass (low level of management) | 40 |
| Grass (intensively managed, using N fertilizer for maximum economic yield) | 70 |
| 2-year stand of red or white clover | 70 |
| 3-year alfalfa stand (20–60% legume) | 70 |
| 3-year alfalfa stand (>60% legume) | 120 |
| Crimson clover | 110 |
| Winter peas | 110 |
| Hairy vetch cover crop (excellent growth) | 110 |
| 1Less credit should be given for sandy soils with high amounts of leaching potential. 2Some labs give 30 or 40 pounds of N credit for soybeans, while others give no N credit. Credits represent the amount of N that will be available to the crop (not the total amount contained in residue). Although the actual amount of N that will become available can be higher in dry years and lower in wet years (Figure 19.2), we still can’t accurately predict the growing season weather. When following cover crops, the stage of growth and the amount of growth will strongly influence the amount of N available to the following crop. | |
Relying on legumes to supply N to following crops. Nitrogen is the only nutrient of which you can “grow” your own supply. High-yielding legume cover crops, such as hairy vetch and crimson clover, can supply most, if not all, of the N needed by the following crop. Growing a legume as a forage crop (alfalfa, alfalfa/ grass, clover, clover/grass) in rotation also can provide much, if not all, of the N for row crops. The N-related aspects of both cover crops and rotations with forages were discussed in chapters 10 and 11.
COVER CROPS ENHANCE P FOR FOLLOWING CROP
Cover crops mobilize and take up a significant amount of P through mycorrhizae and other organisms of the root microbiome. Later, as they decompose, this P becomes available for the following crops to use. While this is a very different mechanism than N fixation by legumes, it is another example of a crop together with microorganisms helping the following crop obtain particular nutrients.
Animals on the farm or on nearby farms? There are many possibilities for actually eliminating the need for N fertilizer if you have ruminant animals on your farm or on nearby farms for which you can grow forage crops (and perhaps use the manure on your farm). A forage legume, such as alfalfa, red clover or white clover, or a grass-legume mix, can supply substantial N for the following crop. Frequently, nutrients are imported onto livestock-based farms as various feeds (usually grains and soybean meal mixes). This means that the manure from the animals will contain nutrients imported from off the farm, and this reduces the need to purchase fertilizers. When planting vegetable crops following a manure application, keep in mind the regulation that requires 120 days from application to harvest (see discussion in Chapter 12 for manure use and food safety issues).
No animals? Although land constraints don’t usually allow it, some vegetable farmers grow a forage legume for one or more years as part of a rotation, even when they are not planning to sell the crop or feed it to animals. They do so to rest the soil and to enhance the soil’s physical and biological properties, and nutrient status. Also, some cover crops, such as hairy vetch—grown off-season in the fall and early spring—can provide sufficient N for some of the high-demanding summer annuals. It’s also possible to undersow sweet clover, planning for fall brassica crops the following year. (If tillage is used, it can be plowed under the next July to prepare for the fall crop.) Sunn hemp and cowpeas growing as cover crops in the Southeast during the summer months have been found to replace one-third to one-half of the N needed for fall broccoli.
Reducing N and P Losses
Manage N and P fertilizers more efficiently. You should have plenty of organic nutrients present if you’ve worked to build and maintain soil organic matter. These readily decomposable fragments provide N and P as they decompose, thereby reducing the amount of fertilizer that’s needed.
When applying commercial fertilizers and manures, the timing and method of application affect the efficiency of use by crops and the amount of loss from soils, especially in humid climates. In general, it is best to apply fertilizers close to the time they are needed by plants, which is especially important when it involves N. Losses of surface-applied fertilizer and manure nutrients are also frequently reduced by soil incorporation with tillage (even a light incorporation can help a lot). Liquid N fertilizer, especially when dribble applied, penetrates the surface, better protecting it from possible gaseous loss. And no-tilled soils that have continual living roots by using cover crops tend to have vastly greater water infiltration and less runoff and gaseous losses.
If you’re growing a crop for which a reliable in-season adaptive method is available, like the PSNT, a sensor or a computer model, you can hold off applying most of the fertilizer until the crop indicates a need. At that point, apply N as a sidedress or topdress. However, if you know that your soil is probably very N deficient (for example, a sandy soil low in organic matter), you may need to band-apply higher-than-normal levels of starter N at planting or broadcast some N before planting to supply sufficient N nutrition until the soil test indicates whether there is a need for more N (applied as a sidedress or topdress). About 15–20 pounds of starter N per acre (in a band at planting) is highly recommended for crops in colder climates. Even more starter N is needed when some cover crops like cereal rye or triticale are allowed to grow near maturity. The large amount of biomass, with its high C:N ratio, will tie up mineral sources of soil N for some weeks following cover crop termination. When organic farmers use fishmeal or seed meals to supply N to crops, they should plan on it becoming available over the season, with little released in the first weeks of decomposition. On the other hand, N contained in feather meal may become available more rapidly.
In-season topdressing N on wheat and on some other annual cereal or oilseed crops is sometimes needed, especially when wet conditions cause significant losses of available soil N. It’s helpful if farmers put high-N strips within fields, in which they apply N at rates of 40–50 pounds per acre higher than other areas. The length and width of the strips aren’t that important. The purpose of the strips is to see if you can tell the difference between the wheat in the high-N strip and the rest of the field. Top dressing N is recommended if the difference is very noticeable.
If the soil is very deficient in phosphorus, P fertilizers have traditionally been incorporated by tillage to raise the general level of the nutrient. Incorporation is not possible with no-till systems, and if a soil is very deficient, some P fertilizer should be incorporated before starting no-till. Nutrients accumulate near the surface of reduced tillage systems when fertilizers or manures are repeatedly surface applied. If P levels are good to start with, in later years small amounts of surface-applied P will work its way deeper into the soil surface. And P can be band applied as starter fertilizer at planting, or it can be injected, keeping it below the surface.
In soils with optimal P levels, some P fertilizer is still recommended, along with N application, for row crops in cool regions. (Potassium is also commonly recommended under these conditions.) Frequently, the soils are cold enough in the spring to slow down root development, P diffusion toward the root and mineralization of P from organic matter, thereby reducing P availability to seedlings. No-tilled soils with plentiful surface residue will stay cool for a longer period in the spring, thereby decreasing both N and P availability. However, if cover crops are used together with no-till—a combination that provides many benefits—soils will dry and warm more rapidly, lessening the concern with early P deficiency in row crops. But for no-till without cover crops in cool climates it is a good idea to use a small amount of starter P for the young crop—even if the soil is in the optimal P soil test range.
Use the right fertilizer products. Some of the N in surface-applied urea, the cheapest and most commonly used solid N fertilizer, is lost as a gas if it is not rapidly incorporated into the soil. If as little as a quarter inch of rain falls within a few days of surface urea application, N losses are usually less than 10%. However, losses may be 30% or more in some cases (a 50% loss may occur following surface application to a calcareous soil that is over pH 8). When urea is used for no-till systems, it can be placed below the surface or surface applied in the form of chemically stabilized urea, greatly reducing N loss. Stabilized urea is the most economical source when N fertilizer is broadcast as a topdress on grass, on cereals such as wheat, or on row crops. Solutions of urea and ammonium nitrate (UAN) are also used as a topdress or are dribbled on as a band. (Although once widely used, solid ammonium nitrate fertilizer is expensive and not always readily available due to concerns about explosivity. But like calcium ammonium nitrate [CAN], its N is generally not lost as a gas when left on the surface and therefore is a good product for topdressing.)
Anhydrous ammonia, the least expensive source of N fertilizer, causes large changes in soil pH in and around the injection band. The pH increases for a period of weeks, many organisms are killed, and organic matter is rendered more soluble. Eventually, the pH decreases, and the band is repopulated by soil organisms. However, significant N losses can occur when anhydrous is applied in a soil that is too dry or too wet. In humid regions, even if stabilizers are used, anhydrous applied long before crop uptake significantly increases the amount of N that may be lost. For this reason, fall-applied anhydrous ammonia is a practical N source only in the more arid western portion of the Corn Belt, and only after the soil has cooled below 50 degrees F. But fall application of anhydrous ammonia remains relatively common even in the more humid parts of the region due to price and logistical benefits, but this raises environmental concerns.
In some cases, nutrients are applied individually through separate fertilizer products, while multi-nutrient compounds (like monoammonium phosphate) or blended materials are used in other cases. When applying multiple nutrients at once, aim to use combinations that proportionally fit the nutritional needs of your crop, thereby reducing unnecessary applications and buildup of nutrients that are overapplied. Or otherwise use multi-nutrient fertilizer in combination with single-nutrient products to achieve the right proportions.
Use nitrogen efficiency enhancement products. Field nitrogen losses can be high depending on the soil, the practices used and the conditions of the growing season, especially weather. With urea-based nitrogen fertilizers and manure, ammonia losses into the atmosphere can be considerable if the material is left on the surface, especially when conditions following application are dry and soil pH is high. Several products on the market reduce ammonia losses by suppressing the activity of the urease enzyme. These urease inhibitors reduce the production of ammonia by naturally occurring soil enzymes, lessening N losses as well as concerns about air pollution and unwanted nitrogen deposition in nearby areas. Nitrification inhibitors are another type of products for use with N fertilizers. These suppress conversion of ammonium to nitrate by naturally occurring soil microorganisms. Ammonium is strongly held by negative charges on soil particles (the cation exchange complex) and does not leach from soils, while the negatively charged nitrate ion can wash through the soil when a lot of rain occurs. This is especially a concern with sandy soils. Also, in finer-textured soils, nitrate can be lost during wet periods through denitrification and volatilization of N2 and N2O into the air. Of course, the leaching and gaseous losses are detrimental to farm profitability as well as to the environment. The role of the nitrification inhibitor is to maintain nitrogen in the ammonium form for longer periods, slowly making nitrate available as the growing crop develops, thereby increasing use efficiency. A third type of product, similar to nitrification inhibitors, focuses on controlled release by using a coating on fertilizer material that causes it to slowly dissolve and release the nitrogen fertilizer.
Soil Reactions with N Fertilizers
Urea is converted to ammonia (lost to the atmosphere or dissolved in water to form ammonium as a gas, or converted to nitrate).
Ammonia and ammonium are nitrified to nitrate (easily lost by leaching and/or denitrification).
The choice of enhanced efficiency products depends on the fertilization strategy. Urease inhibitors are appropriate when using urea-based fertilizers without incorporation. When applying ammonia/ammonium-based fertilizers well before crop uptake, consider adding a nitrification inhibitor or using coated materials. In some cases, a combination of products is appropriate. In general, the use of these products reduces N losses, but it depends on the production environment in a particular growing season. It may prevent yield losses in some years or allow reductions in overall N fertilizer rates by reducing the need for using higher levels of fertilizer as “insurance.”
| Common Nitrogen Fertilizer Efficiency Enhancement Products | ||
|---|---|---|
| Mode of action | Formulation and use | Common enhanced efficiency products1 |
| Urease inhibition | Additive for urea-based; manure | NBPT, MIC |
| Nitrification inhibition | Additive for anhydrous ammonia, urea- and ammonium-based | Nitrapyrin, DCD, MIC |
| Urease and nitrification inhibition | Stand-alone fertilizer product | Ammonium and calcium thiosulfates |
| Controlled release | Stand-alone fertilizer product | Polymer-coated prilled nitrogen or other nutrients |
| 1This list is not comprehensive but includes the most widely used products. Inclusion or omission of a product in this list does not imply an endorsement by the authors or publisher. Source: Cantarella, H., R. Otto, J.R. Soares and A.G. de Brito Silva. 2018. Agronomic efficiency of NBPT as a urease inhibitor: A review. Journal Advanced Research 13: 19–27. | ||
NEW TECHNOLOGY FOR CORN NITROGEN FERTILIZATION
Corn is a tropical plant that is more efficient at utilizing N than are most other crops: it produces more additional yield for each extra pound of N absorbed by the plant. But corn production systems as a whole have low efficiency of fertilizer N, typically less than 50%. Environmental N losses (leaching, denitrification and runoff) are much higher for corn than for crops such as soybeans and wheat, and especially when compared to alfalfa and grasses. This can be attributed to different crop growth cycles, fertilizer rates, fertilizer application schedules, timing of crop water and N uptake, and rooting depths. Intensive corn production areas have therefore become the focus of policy debates that address environmental concerns like groundwater contamination and hypoxia zones in estuaries.
Nitrogen management for corn is still mostly done without recognizing how seasonal weather, particularly precipitation, can cause high N losses through leaching and denitrification. The PSNT was the first approach that addressed these dynamic processes and therefore provided inherently more precise N fertilizer recommendations and eliminated a lot of unnecessary N applications. Still, many farmers like to apply additional “insurance fertilizer” because they want to be certain of an adequate N supply in wet years. But they may actually need it in only, say, one out of four seasons. For those other years, excess N application creates high environmental losses.
New technologies are emerging in addition to the PSNT that allow us to more precisely manage N. Computer models and climate databases can be employed to adapt N recommendations by accounting for weather events and in-field soil variability. Also, crop reflectance of light, which is affected by the degree of N nutrition in the plant, can be measured using aerial and satellite images or tractor-mounted sensors, and can then be used to adjust sidedress N fertilizer rates, even for small zones in a field (precision management).
Use perennial forages (sod-forming crops) in rotations. As we’ve discussed a number of times, rotations that include a perennial forage crop help reduce runoff and erosion; improve beneficial aggregation; break harmful weed, insect and nematode cycles; and build soil organic matter. Decreasing the emphasis on row crops in a rotation and including perennial forages also helps decrease leaching losses of nitrate. This happens for two main reasons:
- There is less water leaching under a sod because it uses more water over the entire growing season than does an annual row crop, which has bare soil in the spring and after harvest in the fall.
- Nitrate concentrations under sod rarely reach anywhere near as high as those under row crops.
So, whether the rotation includes a grass, a legume or a legume-grass mix, the amount of nitrate leaching to groundwater is usually reduced. (A critical step, however, is the conversion from sod to row crop. When a sod crop is plowed, a lot of N is mineralized. If this occurs many months before the row crop takes it up, high nitrate leaching and denitrification losses occur.) Using grass, legume or grass-legume forages in the rotation also helps with P management because of the reduced runoff and erosion, and the effects on soil structure for the following crop.
Use cover (catch) crops to prevent nutrient losses. High levels of soil nitrate may be left at the end of the growing season if drought causes a poor crop year or if excess N fertilizer or manure has been applied. The potential for nitrate leaching and runoff can be significantly reduced if you sow a fast-growing cover crop like cereal rye immediately after the main crop has been harvested. Such cover crops are commonly referred to as “catch crops” because their fast-growing roots can capture the remaining nutrients in the soil and store them in their biomass. One option available to help manage N is to use a combination of a legume and grass. The combination of hairy vetch and cereal rye or triticale works well in cooler temperate regions. When nitrate is scarce, the vetch or crimson clover does much better than the rye, and a large amount of N is fixed for the next crop. Conversely, the rye competes well with the vetch when nitrate is plentiful; less N is fixed (of course, less is needed); and much of the nitrate is tied up in the rye and stored for future use. Crimson clover with either cereal rye or oats works similarly in the South, with the clover growing better and fixing more N when soil nitrate is scarce, and with cereal rye growing faster when nitrate is plentiful.
In general, having any cover crop on the soil during the off-season is helpful for P management. A cover crop that establishes quickly and helps protect the soil against erosion will help reduce P losses.
Reduce tillage. Because most P is lost from fields by sediment erosion, environmentally sound P management should include reduced tillage systems. Leaving residues on the surface and maintaining stable soil aggregation and lots of large pores help water to infiltrate into soils. When runoff does occur, less sediment is carried along with it than when conventional plow-harrow tillage is used. Reduced tillage, by decreasing runoff and erosion, usually decreases both P and N losses from fields. Recent studies have also shown that reduced tillage results in more effective N cycling. Although N fertilizer needs are generally slightly higher in early transition years, long-term no-till increases organic matter contents over conventional tillage and also, after some years, results in 30 pounds (or more) per acre greater N mineralization, which is a significant economic benefit to the farm.
TILLAGE, NUTRIENT LOSS, AND FERTILIZER APPLICATION METHODS
Reducing tillage usually leads to marked reductions of nitrate leaching loss to groundwater as well as to runoff and, therefore, N and P loss in runoff. But, questions have come up about potential problems with broadcasting N and P fertilizers in reduced tillage systems, especially in no-till. The main attractiveness of broadcast fertilizer is that you can travel faster and cover more land than with injection methods of application—around 500–800 acres in eight hours for broadcast versus about 200 acres for injection. However, there are two complicating factors.
- If intense storms occur soon after application of surface-applied urea, N is more likely to be lost via leaching than if it had been incorporated. Much of the water will flow over the surface of no-till soils, picking up nitrate and urea, before entering wormholes and other channels. It then easily moves deep into the subsoil. It is best not to broadcast N fertilizer and to leave it on the surface with a no-till system. This is particularly true for urea, since surface residues contain higher levels of the urease enzyme, facilitating fast conversion to ammonia, which is rapidly lost as a gas. Fertilizer N may be applied at different stages: before planting, with the seed at planting, or as a sidedress. Using liquid N as a sidedress results in better soil contact than a solid fertilizer would achieve.
P accumulates on the surface of no-till soils (because there is no incorporation of broadcast fertilizers, manures, crop residues or cover crops). Although there is usually less runoff, fewer sediments and less total P lost with no-till, the concentration of dissolved P in the runoff is often higher than for conventionally tilled soils. Phosphorus should be applied below the surface to reduce such losses.
Working Toward Balancing Nutrient Imports and Exports
In addition to being contained in the products sold off the farm, nitrogen and phosphorus are lost from soils in many unintended ways, including runoff that takes both N and P, nitrate leaching (and in some situations, P as well), denitrification, and volatilization of ammonia from surface-applied urea and manures. Even if you take all precautions to reduce unnecessary losses, some N and P loss will occur. While you can easily overdo it with fertilizers, using more N and P than is needed also occurs on many livestock farms that import a significant proportion of their feeds. If a forage legume, such as alfalfa, is an important part of the rotation, the combination of biological N fixation plus imported N in feeds may exceed the farm’s needs. A reasonable goal for farms with a large net inflow of N and P through feed would be to try to reduce imports of these nutrients onto the farm (including legume N), or to increase exports, to a point closer to balance.
On crop farms, as well as on livestock-based farms with low numbers of animals per acre, it’s fairly easy to bring inflows and outflows into balance by properly crediting N from the previous crop, and N and P in manure. But it is a more challenging problem when there are a large number of animals for a fixed land base and a large percentage of the feed must be imported. This happens frequently on factory-type animal production facilities, but it can also happen on smaller, family-sized farms. At some point, thought needs to be given to either expanding the farm’s land base or exporting some of the manure to other farms. In the Netherlands, nutrient accumulation on livestock farms became a national problem and generated legislation that limits animal units on farms. One option is to compost the manure, which makes it easier to transport or sell. It causes some N losses during the composting process, but stabilizes the remaining N before application. On the other hand, the availability of P in manure is not greatly affected by composting. That’s why using compost to supply a particular amount of “available” N usually results in applications of larger total amounts of P than plants need.
Using Organic Sources of Phosphorus and Potassium
Manures and other organic amendments are frequently applied to soils at rates estimated to satisfy a crop’s N need. This commonly adds more P and potassium than the crop needs. After many years of continuous application of these sources to meet N needs, soil test levels for P and potassium may be in the excessive range. Although there are a number of ways to deal with this issue, all solutions require reduced applications of fertilizer P and P-containing organic amendments. If it’s a farm-wide problem, some manure may need to be exported and N fertilizer or legumes relied on to provide N to grain crops. Sometimes, it’s just a question of better distribution of manure around the various fields: getting to those fields far from the barn more regularly. Changing the rotation to include crops such as alfalfa, for which no manure N is needed, can help. However, if you’re raising livestock on a limited land base, you should make arrangements to have the manure used on a neighboring farm or sell the manure to a composting facility.
Managing High-P Soils
High-P soils occur because of a history of either excessive applications of P fertilizers or, more commonly, application of lots of manure. This is a problem on livestock farms with limited land and where a medium-to-high percentage of feed is imported. The nutrients imported in feeds may greatly exceed the nutrients exported in animal products. In addition, where manures or composts are used at recommended rates for providing sufficient N to crops, more P than needed usually is added. It’s probably a good idea to reduce the potential for P loss from all high-P soils. However, it is especially important to reduce the risk of environmental harm from those high-P soils that are also likely to produce significant runoff (because of slope, fine texture, poor structure or poor drainage). Therefore, the environmental context should be considered. If the farm is near a critical water resource that is impacted by field runoff or tile drainage, aggressive measures are needed to reduce the impact. Conversely, small vegetable operations or urban farms on flat ground where fields are surrounded by grass berms or alleyways pose much lower risk, and high soil-P levels are generally more acceptable.
There are a number of practices that should be followed with high-P soils.
- First, deal with the “front end” and reduce animal P intake to the lowest levels needed. Not that long ago a survey found that the average dairy herd in the United States was fed about 25% more P than recommended by the standard authority (the National Research Council, or NRC). Using so much extra can cost dairy farmers thousands of dollars to feed a 100-cow herd supplemental P that the animals don’t need and that ends up as a potential pollutant.
- Second, reduce or eliminate applications of extra P. For a livestock farm, this may mean obtaining the use of more land to grow crops, to spread manure over a larger land area, or to swap fields with nearby farms that don’t have high-P problems. For a crop farm, this may mean using legume cover crops and forages in rotations to supply N without adding P. The cover crops and forage rotation crops are also helpful to build up and maintain good organic matter levels in the absence of importing manures or composts, or other organic material from off the farm. The lack of imported organic sources of nutrients (to try to reduce P imports) means that a crop farmer will need to be more creative using crop residues, rotations and cover crops to maintain good organic matter levels. Also, don’t use a high-P source to meet N demands. Compost has many benefits, but if used to provide N fertility, it will build up P over the long term.
- Third, reduce runoff and erosion to minimal levels. P is usually a problem only if it gets into surface waters. Anything that helps water infiltration or impedes water and sediments from leaving the field—reduced tillage, strip cropping along the contour, cover crops, grassed waterways, riparian buffer strips, etc.—decreases problems caused by high-P soils. (Note: Significant P losses in tile drainage water have been observed, especially from fields where large amounts of liquid manure are applied.)
- Fourth, continue to monitor soil P levels. Soil-test P will slowly decrease over the years once P imports, in the form of fertilizers, organic amendments or feeds, are reduced or eliminated. Soils should be tested every two to three years for other reasons, anyway. So just remember to keep track of soil-test P to confirm that levels are decreasing. Phosphorus accumulates especially rapidly in the surface of no-till soils that have received large applications of manure or fertilizer over the years. One management option in these cases is a one-time tillage of the soil to incorporate the high-P soil layer. If this is done, use practices that don’t result in building up surface soil P once again, such as applying P as starter near the seed and injection (especially liquid manure) instead of broadcast applications.
Chapter 19 Summary
Both N and P are needed by plants in large amounts, but when soils are too rich in these nutrients, they are environmental hazards. And although N and P behave somewhat differently in soils, most sound management practices for one are also sound for the other. Using soil tests, comprehensive nutrient management planning and recommendation tools that account for all sources, such as soil organic matter, manures, cover crops and decomposing sods, can help better manage these nutrients. Reduced tillage, cover crops and rotation with sod crops decrease runoff and erosion and help in many other ways, including better N and P management. In addition, following the 4R-Plus principles and using technologies like N stabilizers/inhibitors as well as sensors and models can increase the use efficiency of N and P, and can reduce detrimental environmental impacts.
Chapter 19 Sources
Balkcom, K.S., A.M. Blackmer, D.J. Hansen, T.F. Morris and A.P. Mallarino. 2003. Testing soils and cornstalks to evaluate nitrogen management on the watershed scale. Journal of Environmental Quality 32: 1015–1024.
Brady, N.C. and R.R. Weil. 2008. The Nature and Properties of Soils, 14th ed. Prentice Hall: Upper Saddle River, NJ.
Cassman, K.G., A. Dobermann and D.T. Walters. 2002. Agroecosystems, nitrogen-use efficiency, and nitrogen management. Ambio 31: 132–140.
Jokela, B., F. Magdoff, R. Bartlett, S. Bosworth and D. Ross. 1998. Nutrient Recommendations for Field Crops in Vermont. University of Vermont, Extension Service: Burlington, VT.
Kay, B.D., A.A. Mahboubi, E.G. Beauchamps and R.S. Dharmakeerthi. 2006. Integrating soil and weather data to describe variability in plant available nitrogen. Soil Science Society of America Journal 70: 1210–1221.
Laboski, C.A.M., J.E. Sawyer, D.T. Walters, L.G. Bundy, R.G. Hoeft, G.W. Randall and T.W. Andraski. 2008. Evaluation of the Illinois Soil Nitrogen Test in the North Central region of the United States. Agronomy Journal 100: 1070–1076.
Lazicki, P., D. Geisseler and M. Lloyd. 2020. Nitrogen mineralization from organic amendments is variable but predictable. Journal of Environmental Quality 49:483–495.
Magdoff, F.R. 1991. Understanding the Magdoff pre-sidedress nitrate soil test for corn. Journal of Production Agriculture 4: 297–305.
Mitsch, W.J., J.W. Day, J.W. Gilliam, P.M. Groffman, D.L. Hey, G.W. Randall and N. Wang. 2001. Reducing nitrogen loading to the Gulf of Mexico from the Mississippi River basin: Strategies to counter a persistent ecological problem. BioScience 51: 373–388.
Morris, T.F., T. Scott Murrell, Douglas B. Beegle, James J. Camberato, Richard B. Ferguson, John Grove, Quirine Ketterings, Peter M. Kyveryga, Carrie A.M. Laboski, Joshua M. McGrath, John J. Meisinger, Jeff Melkonian, Bianca N. Moebius-Clune, Emerson D. Nafziger, Deanna Osmond, John E. Sawyer, Peter C. Scharf, Walter Smith, John T. Spargo, Harold M. van Es and Haishun Yang. 2018. Strengths and Limitations of Nitrogen Rate Recommendations for Corn and Opportunities for Improvement. Agron. J. 110:1–37.
National Research Council. 1988. Nutrient Requirements of Dairy Cattle, 6th rev. Ed. National Academy Press: Washington, DC.
Olness, A.E., D. Lopez, J. Cordes, C. Sweeney and W.B. Voorhees. 1998. Predicting nitrogen fertilizer needs using soil and climatic data. In Procedures of the 11th World Fertilizer Congress, Gent, Belgium, Sept. 7–13, 1997, ed. A. Vermoesen, pp. 356–364. International Centre of Fertilizers: Gent, Belgium.
Sawyer, J., E. Nafziger, G. Randall, L. Bundy, G. Rehm and B. Joern. 2006. Concepts and Rationale for Regional Nitrogen Guidelines for Corn. Iowa State University Extension Publication PM2015, 27 pp.
Sela. S. and H.M. van Es. 2018. Dynamic tools unify fragmented 4Rs into an integrative nitrogen management approach. J Soil & Water Conserv. 73:107A–112A.
Sharpley, A.N. 1996. Myths about P. Proceedings from the Animal Agriculture and the Environment North American Conference, Dec. 11–13, Rochester, NY. Northeast Region Agricultural Engineering Service: Ithaca, NY.
Vigil, M.F. and D.E. Kissel. 1991. Equations for estimating the amount of nitrogen mineralized from crop residues. Soil Science Society of America Journal 55: 757–761. Wortmann, C., M. Helmers, A. Mallarino, C. Barden, D. Devlin, G. Pierzynski, J. Lory, R. Massey, J. Holz and C. Shapiro. 2005. Agricultural Phosphorus Management and Water Quality Protection in the Midwest. University of Nebraska: Lincoln, NE.
